Written by Caroline Anderson
Edited by William Foster and Arunon Sivananthan
This week we were fortunate enough to welcome a world-renowned biomedical scientist to the IRD journal club. Professor Jeffrey Pollard is currently the Director of the Medical Research Council Centre for Reproductive Health at the University of Edinburgh. His seminar earlier in the day had focussed on the role of the tumour microenvironment, and metastasis-associated macrophages (MAMs) in particular, in breast cancer metastasis. Much of the work he discussed can be found here: Kitamura et al, JEM, 2015 (PMID: 26056232). 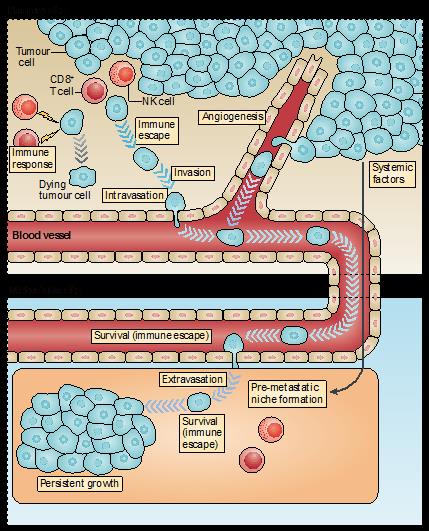
Both pro- and anti-tumorigenic immune cells are found in association with tumours. Indeed, it appears that tumour-associated macrophages can be influenced by the tumour microenvironment into adopting a more tumour-promoting phenotype (Kitamura, Qian and Pollard, Nature Reviews Immunology, 2015). Previous work showed that antibody blockade of CCL2 signalling inhibited the recruitment of inflammatory monocytes (IMs) and reduced MAM numbers in lungs with metastatic tumours (Qian et al, 2009). As such, Kitamura et al set out to test the hypothesis that the CCL2-CCR2 axis was involved in regulating MAM function and MAM-mediated metastasis of cancer cells.
Firstly, they performed a microarray to identify potential downstream targets of CCR2 signalling in MAMs. Interestingly, Ccl3 expression was significantly downregulated in macrophages lacking CCR2 when compared to wild-type macrophages. Secretion of CCL3 increased when wild-type but not CCR2-deficient macrophages were stimulated with recombinant CCL2. Hence, CCL2 signalling via CCR2 can increase CCL3 secretion from macrophages. This chemokine-induced chemokine secretion was also apparent when human monocyte-derived macrophages were stimulated with recombinant human CCL2 or conditioned medium from human breast cancer cells, whilst the effect was abolished in the presence of anti-CCL2.
The Pollard lab make extensive use of a genetically engineered mouse model of breast cancer called the Polyoma Middle T mouse model. In this model, the Polyoma Middle T oncogene is expressed specifically in the mammary epithelium, leading to spontaneous metastases which preferentially migrate to the lung. Using this model, Kitamura et al show that genetic deletion of CCL3 or CCR1 in macrophages caused a decrease in the number of metastatic foci and reduced MAM numbers in the lung. Conversely, adoptive transfer of CCR3+ IMs to Ccl3-deficient mice increased metastatic seeding of cancer cells.
The CCL3 receptors, CCR1 and CCR5, are predominantly expressed by macrophages rather than breast cancer or endothelial cells, suggesting that CCL3 acts in an autocrine manner on MAMs. Using CMFDA-labelled inflammatory monocytes, the authors found that CCR1 deficiency affected the retention of MAMs in the metastatic lung but not the recruitment or differentiation of IMs to the lung. Finally, the interaction between tumour cells and MAMs was found to be dependent on a4-integrin expression by leukocytes and expression of its ligand, VCAM-1 by breast cancer cells. Taken together, the paper concludes that a CCL2-induced signalling cascade in macrophages leads to enhanced metastatic seeding of breast cancer cells through the increased retention of MAMs.
The journal club discussion centred around Professor Pollard’s experiences of working in the US and the opportunities afforded by working abroad. Moving back to science, we touched on the intriguing evidence that chemokines play roles above and beyond chemotaxis, which could have consequences for the therapeutic targeting of these signalling molecules in disease.